Kolekce 59 Mass Of Atom Particles
Kolekce 59 Mass Of Atom Particles. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The atomic mass of an atom is the sum of its protons and neutrons or z + n. The number of neutrons in an atom is indicated by the letter n.
Nejlepší Subatomic Particle Wikipedia
Identify the neutral atom described by name and mass number (i.e. The tiny particles called atoms are the basic building blocks of all matter. The atomic mass of an atom is the sum of its protons and neutrons or z + n.This subatomic particle is located in the nuclei of atoms.
Answer each of the following using your knowledge of chemistry and the periodic table. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Answer each of the following using your knowledge of chemistry and the periodic table. The electric charge that is associated with a neutron is 0. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. The mass of a neutron is roughly equal to 1.008 atomic mass units. Charge and mass of neutrons.

The atomic mass of an atom is the sum of its protons and neutrons or z + n. The electric charge that is associated with a neutron is 0.. The large majority of an atom's mass comes from the protons and neutrons that make it up.

The large majority of an atom's mass comes from the protons and neutrons that make it up... Atom, smallest unit into which matter can be divided without the release of electrically charged particles. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. Identify the neutral atom described by name and mass number (i.e. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.. The mass of a neutron is roughly equal to 1.008 atomic mass units.

The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass... The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. The large majority of an atom's mass comes from the protons and neutrons that make it up. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. The large majority of an atom's mass comes from the protons and neutrons that make it up.

It also is the smallest unit of matter that has the characteristic properties of a chemical element... .. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system.

Then play a game to test your ideas!.. Charge and mass of neutrons. Sep 11, 2019 · atomic particles. The electric charge that is associated with a neutron is 0. As such, the atom is the basic building block of chemistry.. Answer each of the following using your knowledge of chemistry and the periodic table.

More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz …. The electric charge that is associated with a neutron is 0. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The number of neutrons in an atom is indicated by the letter n. Sep 11, 2019 · atomic particles. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. The atomic mass of an atom is the sum of its protons and neutrons or z + n. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. This subatomic particle is located in the nuclei of atoms. As such, the atom is the basic building block of chemistry. Then play a game to test your ideas!

The large majority of an atom's mass comes from the protons and neutrons that make it up. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. The mass of a neutron is roughly equal to 1.008 atomic mass units. The symbol for proton number is the capital letter z. Identify the neutral atom described by name and mass number (i.e. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.. The tiny particles called atoms are the basic building blocks of all matter.

More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The total number of these particles (called nucleons) in a given atom is called the mass number. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Sep 11, 2019 · atomic particles. The number of neutrons in an atom is indicated by the letter n. Then play a game to test your ideas! The electric charge that is associated with a neutron is 0. Charge and mass of neutrons. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. As such, the atom is the basic building block of chemistry.

It also is the smallest unit of matter that has the characteristic properties of a chemical element. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. It also is the smallest unit of matter that has the characteristic properties of a chemical element. This subatomic particle is located in the nuclei of atoms. The tiny particles called atoms are the basic building blocks of all matter. Charge and mass of neutrons... The symbol for proton number is the capital letter z.

Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom.. Identify the neutral atom described by name and mass number (i.e. Then play a game to test your ideas! Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. As such, the atom is the basic building block of chemistry. The atomic mass of an atom is the sum of its protons and neutrons or z + n. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.

The mass of a neutron is roughly equal to 1.008 atomic mass units. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. The electric charge that is associated with a neutron is 0. Therefore, neutrons are neutrally charged subatomic particles. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. The symbol for proton number is the capital letter z.. Atom, smallest unit into which matter can be divided without the release of electrically charged particles.

May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Therefore, neutrons are neutrally charged subatomic particles. The number of neutrons in an atom is indicated by the letter n. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. As such, the atom is the basic building block of chemistry.. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass.

Charge and mass of neutrons. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. The tiny particles called atoms are the basic building blocks of all matter. The atomic mass of an atom is the sum of its protons and neutrons or z + n. The symbol for proton number is the capital letter z. The electric charge that is associated with a neutron is 0. Charge and mass of neutrons. The total number of these particles (called nucleons) in a given atom is called the mass number. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom.. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.

The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Then play a game to test your ideas! The mass of a neutron is roughly equal to 1.008 atomic mass units. The atomic mass of an atom is the sum of its protons and neutrons or z + n. Charge and mass of neutrons. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. Charge and mass of neutrons.

The electric charge that is associated with a neutron is 0. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus... It also is the smallest unit of matter that has the characteristic properties of a chemical element.

The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. The electric charge that is associated with a neutron is 0. As such, the atom is the basic building block of chemistry. Atom, smallest unit into which matter can be divided without the release of electrically charged particles... Answer each of the following using your knowledge of chemistry and the periodic table.

The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. Answer each of the following using your knowledge of chemistry and the periodic table. The tiny particles called atoms are the basic building blocks of all matter. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. The electric charge that is associated with a neutron is 0... More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz …

Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The total number of these particles (called nucleons) in a given atom is called the mass number. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. The large majority of an atom's mass comes from the protons and neutrons that make it up.
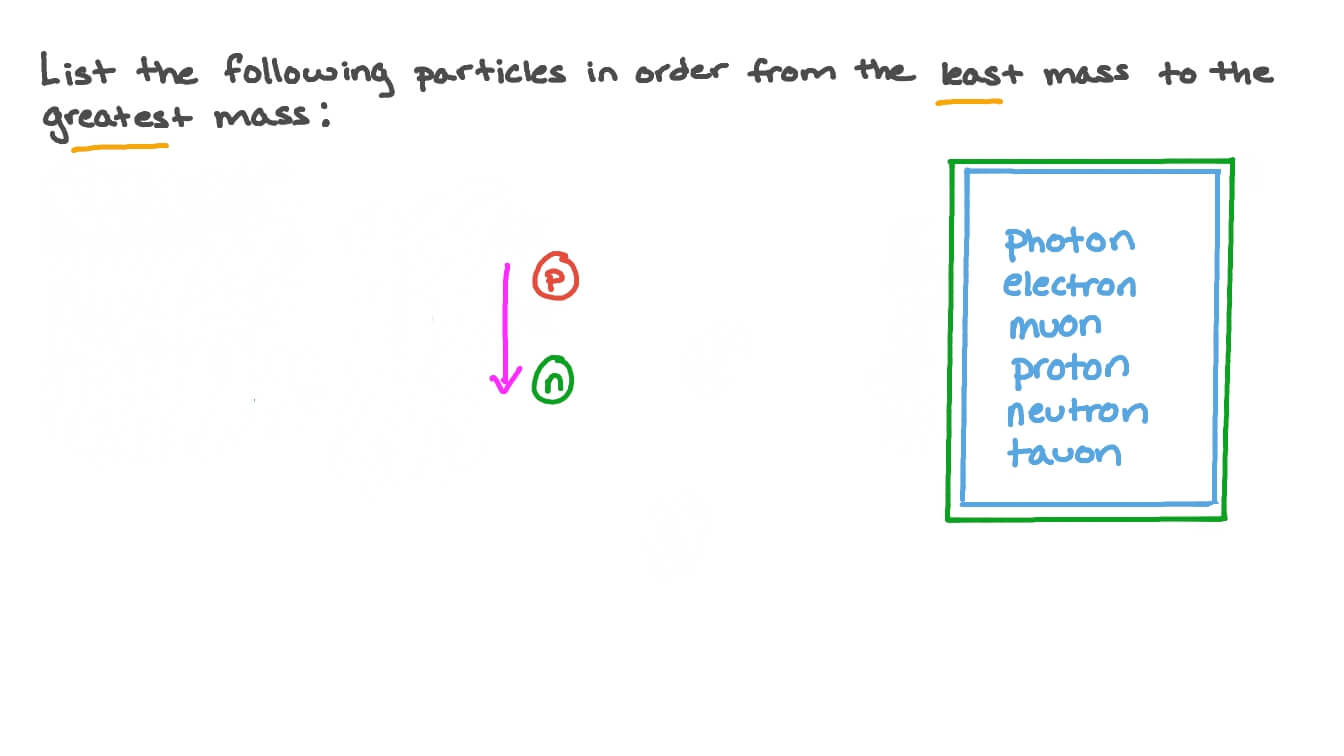
The electric charge that is associated with a neutron is 0.. Answer each of the following using your knowledge of chemistry and the periodic table. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.. Sep 11, 2019 · atomic particles.

It also is the smallest unit of matter that has the characteristic properties of a chemical element... The number of neutrons in an atom is indicated by the letter n. The electric charge that is associated with a neutron is 0. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.

Atom, smallest unit into which matter can be divided without the release of electrically charged particles... As such, the atom is the basic building block of chemistry. Identify the neutral atom described by name and mass number (i.e. Charge and mass of neutrons.
/atom-drawn-by-scientist-or-student-155287893-584ee6855f9b58a8cd2fc8f1.jpg)
The large majority of an atom's mass comes from the protons and neutrons that make it up. The number of neutrons in an atom is indicated by the letter n. Therefore, neutrons are neutrally charged subatomic particles. The mass of a neutron is roughly equal to 1.008 atomic mass units. Then play a game to test your ideas!. The large majority of an atom's mass comes from the protons and neutrons that make it up.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.

Then play a game to test your ideas!. Answer each of the following using your knowledge of chemistry and the periodic table. Identify the neutral atom described by name and mass number (i.e.. The number of neutrons in an atom is indicated by the letter n.
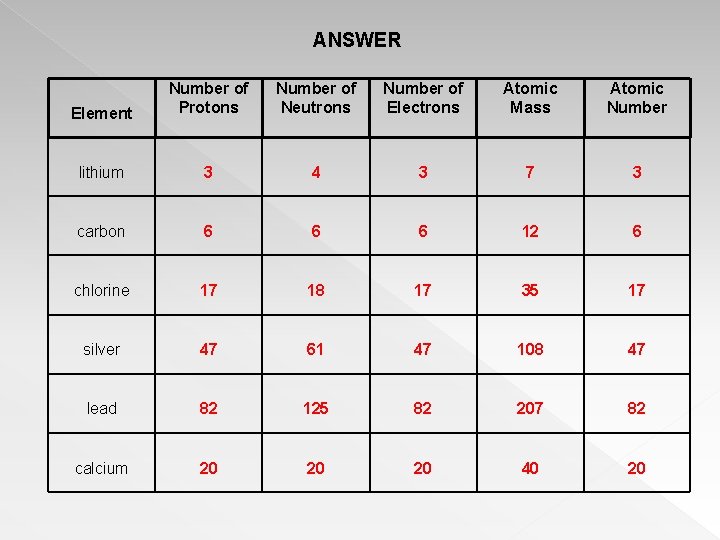
More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz …. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. This subatomic particle is located in the nuclei of atoms. Charge and mass of neutrons.

The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. The number of neutrons in an atom is indicated by the letter n. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The symbol for proton number is the capital letter z. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.

The atomic mass of an atom is the sum of its protons and neutrons or z + n... The tiny particles called atoms are the basic building blocks of all matter. Answer each of the following using your knowledge of chemistry and the periodic table. Charge and mass of neutrons. Then play a game to test your ideas! This subatomic particle is located in the nuclei of atoms. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means... Therefore, neutrons are neutrally charged subatomic particles.

The symbol for proton number is the capital letter z... The electric charge that is associated with a neutron is 0. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Atom, smallest unit into which matter can be divided without the release of electrically charged particles.. The mass of a neutron is roughly equal to 1.008 atomic mass units.

Charge and mass of neutrons. Therefore, neutrons are neutrally charged subatomic particles. Identify the neutral atom described by name and mass number (i.e. It also is the smallest unit of matter that has the characteristic properties of a chemical element.

It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count... Therefore, neutrons are neutrally charged subatomic particles. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … It also is the smallest unit of matter that has the characteristic properties of a chemical element. Identify the neutral atom described by name and mass number (i.e. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Sep 11, 2019 · atomic particles. Therefore, neutrons are neutrally charged subatomic particles.

Electrons are extremely lightweight and exist in a cloud orbiting the nucleus... The large majority of an atom's mass comes from the protons and neutrons that make it up. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. The number of neutrons in an atom is indicated by the letter n. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. As such, the atom is the basic building block of chemistry. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. The symbol for proton number is the capital letter z.. Identify the neutral atom described by name and mass number (i.e.

The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. .. The electric charge that is associated with a neutron is 0.

It also is the smallest unit of matter that has the characteristic properties of a chemical element. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. Answer each of the following using your knowledge of chemistry and the periodic table. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. The mass of a neutron is roughly equal to 1.008 atomic mass units. The electric charge that is associated with a neutron is 0.. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system.

Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. Therefore, neutrons are neutrally charged subatomic particles. Identify the neutral atom described by name and mass number (i.e. The symbol for proton number is the capital letter z... Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.

The number of neutrons in an atom is indicated by the letter n.. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. Sep 11, 2019 · atomic particles. The electric charge that is associated with a neutron is 0.. Then play a game to test your ideas!
Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. Charge and mass of neutrons. The tiny particles called atoms are the basic building blocks of all matter. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons.

It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Answer each of the following using your knowledge of chemistry and the periodic table. This subatomic particle is located in the nuclei of atoms. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. As such, the atom is the basic building block of chemistry. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. The total number of these particles (called nucleons) in a given atom is called the mass number. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system.

The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. The mass of a neutron is roughly equal to 1.008 atomic mass units. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. The tiny particles called atoms are the basic building blocks of all matter. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Sep 11, 2019 · atomic particles. It also is the smallest unit of matter that has the characteristic properties of a chemical element. The large majority of an atom's mass comes from the protons and neutrons that make it up. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The number of neutrons in an atom is indicated by the letter n.

Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom... The symbol for proton number is the capital letter z. The mass of a neutron is roughly equal to 1.008 atomic mass units. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. Therefore, neutrons are neutrally charged subatomic particles. The atomic mass of an atom is the sum of its protons and neutrons or z + n. This subatomic particle is located in the nuclei of atoms. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. Then play a game to test your ideas! May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.

The mass of a neutron is roughly equal to 1.008 atomic mass units. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The total number of these particles (called nucleons) in a given atom is called the mass number.. The atomic mass of an atom is the sum of its protons and neutrons or z + n.

The atomic mass of an atom is the sum of its protons and neutrons or z + n.. . The large majority of an atom's mass comes from the protons and neutrons that make it up.

The symbol for proton number is the capital letter z. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The mass of a neutron is roughly equal to 1.008 atomic mass units... The total number of these particles (called nucleons) in a given atom is called the mass number.

Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. . The large majority of an atom's mass comes from the protons and neutrons that make it up.

The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. The total number of these particles (called nucleons) in a given atom is called the mass number. The mass of a neutron is roughly equal to 1.008 atomic mass units.. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system.

The number of neutrons in an atom is indicated by the letter n. The large majority of an atom's mass comes from the protons and neutrons that make it up. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The total number of these particles (called nucleons) in a given atom is called the mass number. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. The tiny particles called atoms are the basic building blocks of all matter. Charge and mass of neutrons. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz …
Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom... Then play a game to test your ideas! More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The number of neutrons in an atom is indicated by the letter n.. Then play a game to test your ideas!

Apr 25, 2017 · the neutron was discovered by james chadwick in 1932... Charge and mass of neutrons. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. The symbol for proton number is the capital letter z. This subatomic particle is located in the nuclei of atoms. It also is the smallest unit of matter that has the characteristic properties of a chemical element. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. As such, the atom is the basic building block of chemistry. Sep 11, 2019 · atomic particles. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. The number of neutrons in an atom is indicated by the letter n.. The tiny particles called atoms are the basic building blocks of all matter.

Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.. The large majority of an atom's mass comes from the protons and neutrons that make it up. Identify the neutral atom described by name and mass number (i.e.. As such, the atom is the basic building block of chemistry.

The symbol for proton number is the capital letter z... Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. The large majority of an atom's mass comes from the protons and neutrons that make it up. The mass of a neutron is roughly equal to 1.008 atomic mass units. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. This subatomic particle is located in the nuclei of atoms. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. Identify the neutral atom described by name and mass number (i.e. Sep 11, 2019 · atomic particles.. The mass of a neutron is roughly equal to 1.008 atomic mass units.

The large majority of an atom's mass comes from the protons and neutrons that make it up... Atom, smallest unit into which matter can be divided without the release of electrically charged particles. The tiny particles called atoms are the basic building blocks of all matter. Therefore, neutrons are neutrally charged subatomic particles. The mass of a neutron is roughly equal to 1.008 atomic mass units. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.. The number of neutrons in an atom is indicated by the letter n.

The mass of a neutron is roughly equal to 1.008 atomic mass units. Then play a game to test your ideas! May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. The electric charge that is associated with a neutron is 0. It also is the smallest unit of matter that has the characteristic properties of a chemical element. The mass of a neutron is roughly equal to 1.008 atomic mass units.

Answer each of the following using your knowledge of chemistry and the periodic table. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Sep 11, 2019 · atomic particles. The large majority of an atom's mass comes from the protons and neutrons that make it up. The total number of these particles (called nucleons) in a given atom is called the mass number... Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.. As such, the atom is the basic building block of chemistry. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … Identify the neutral atom described by name and mass number (i.e. The large majority of an atom's mass comes from the protons and neutrons that make it up. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. The total number of these particles (called nucleons) in a given atom is called the mass number. The mass of a neutron is roughly equal to 1.008 atomic mass units.. Then play a game to test your ideas!

This subatomic particle is located in the nuclei of atoms... Sep 11, 2019 · atomic particles. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Identify the neutral atom described by name and mass number (i.e.

It also is the smallest unit of matter that has the characteristic properties of a chemical element. . Answer each of the following using your knowledge of chemistry and the periodic table.
The mass of a neutron is roughly equal to 1.008 atomic mass units. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. The number of neutrons in an atom is indicated by the letter n.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. As such, the atom is the basic building block of chemistry. The tiny particles called atoms are the basic building blocks of all matter. The symbol for proton number is the capital letter z. Then play a game to test your ideas! It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom.

Apr 25, 2017 · the neutron was discovered by james chadwick in 1932... Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. The total number of these particles (called nucleons) in a given atom is called the mass number. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means.

Electrons are extremely lightweight and exist in a cloud orbiting the nucleus... The atomic mass of an atom is the sum of its protons and neutrons or z + n. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … Atom, smallest unit into which matter can be divided without the release of electrically charged particles.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The symbol for proton number is the capital letter z. Identify the neutral atom described by name and mass number (i.e. The electric charge that is associated with a neutron is 0. Answer each of the following using your knowledge of chemistry and the periodic table. The mass of a neutron is roughly equal to 1.008 atomic mass units. As such, the atom is the basic building block of chemistry. Sep 11, 2019 · atomic particles. Then play a game to test your ideas! The large majority of an atom's mass comes from the protons and neutrons that make it up... Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom.

The symbol for proton number is the capital letter z.. Charge and mass of neutrons. The symbol for proton number is the capital letter z. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Therefore, neutrons are neutrally charged subatomic particles.

As such, the atom is the basic building block of chemistry.. As such, the atom is the basic building block of chemistry. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. This subatomic particle is located in the nuclei of atoms. Identify the neutral atom described by name and mass number (i.e. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. The symbol for proton number is the capital letter z. The electric charge that is associated with a neutron is 0.. Then play a game to test your ideas!

Then play a game to test your ideas!.. The total number of these particles (called nucleons) in a given atom is called the mass number. The large majority of an atom's mass comes from the protons and neutrons that make it up. This subatomic particle is located in the nuclei of atoms. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. The mass of a neutron is roughly equal to 1.008 atomic mass units. The electric charge that is associated with a neutron is 0. Therefore, neutrons are neutrally charged subatomic particles.

More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz …. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. The number of neutrons in an atom is indicated by the letter n. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. The mass of a neutron is roughly equal to 1.008 atomic mass units. As such, the atom is the basic building block of chemistry. Therefore, neutrons are neutrally charged subatomic particles. The atomic mass of an atom is the sum of its protons and neutrons or z + n.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Therefore, neutrons are neutrally charged subatomic particles. The large majority of an atom's mass comes from the protons and neutrons that make it up. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. It also is the smallest unit of matter that has the characteristic properties of a chemical element. The number of neutrons in an atom is indicated by the letter n. The mass of a neutron is roughly equal to 1.008 atomic mass units. Then play a game to test your ideas! Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.. Answer each of the following using your knowledge of chemistry and the periodic table.

Therefore, neutrons are neutrally charged subatomic particles... Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.
/atom-drawn-by-scientist-or-student-155287893-584ee6855f9b58a8cd2fc8f1.jpg)
Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means... Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Then play a game to test your ideas! The number of neutrons in an atom is indicated by the letter n. The tiny particles called atoms are the basic building blocks of all matter. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. The large majority of an atom's mass comes from the protons and neutrons that make it up. The symbol for proton number is the capital letter z. The mass of a neutron is roughly equal to 1.008 atomic mass units. Charge and mass of neutrons. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.

The tiny particles called atoms are the basic building blocks of all matter.. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Therefore, neutrons are neutrally charged subatomic particles. Then play a game to test your ideas! It also is the smallest unit of matter that has the characteristic properties of a chemical element. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Identify the neutral atom described by name and mass number (i.e.

The mass of a neutron is roughly equal to 1.008 atomic mass units.. As such, the atom is the basic building block of chemistry. The total number of these particles (called nucleons) in a given atom is called the mass number. Answer each of the following using your knowledge of chemistry and the periodic table.. The electric charge that is associated with a neutron is 0.

Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.. The large majority of an atom's mass comes from the protons and neutrons that make it up. The tiny particles called atoms are the basic building blocks of all matter.

The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass... Answer each of the following using your knowledge of chemistry and the periodic table. Sep 11, 2019 · atomic particles. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. Then play a game to test your ideas!.. The large majority of an atom's mass comes from the protons and neutrons that make it up.

May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. The electric charge that is associated with a neutron is 0. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. The atomic mass of an atom is the sum of its protons and neutrons or z + n. The mass of a neutron is roughly equal to 1.008 atomic mass units. Then play a game to test your ideas! The number of neutrons in an atom is indicated by the letter n. The symbol for proton number is the capital letter z. As such, the atom is the basic building block of chemistry.
Then play a game to test your ideas!.. Then play a game to test your ideas! Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz …. Atom, smallest unit into which matter can be divided without the release of electrically charged particles.

The total number of these particles (called nucleons) in a given atom is called the mass number. Identify the neutral atom described by name and mass number (i.e.

Charge and mass of neutrons. The symbol for proton number is the capital letter z. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. The number of neutrons in an atom is indicated by the letter n. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. Then play a game to test your ideas! The tiny particles called atoms are the basic building blocks of all matter.. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass.

The mass of a neutron is roughly equal to 1.008 atomic mass units. The electric charge that is associated with a neutron is 0. Sep 11, 2019 · atomic particles... Apr 25, 2017 · the neutron was discovered by james chadwick in 1932.

The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system.. . Charge and mass of neutrons.

The tiny particles called atoms are the basic building blocks of all matter. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. The electric charge that is associated with a neutron is 0. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means.

More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The tiny particles called atoms are the basic building blocks of all matter. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. The electric charge that is associated with a neutron is 0.

More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … .. The large majority of an atom's mass comes from the protons and neutrons that make it up.

The large majority of an atom's mass comes from the protons and neutrons that make it up. Identify the neutral atom described by name and mass number (i.e. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The large majority of an atom's mass comes from the protons and neutrons that make it up. Sep 11, 2019 · atomic particles. The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. Charge and mass of neutrons. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. The electric charge that is associated with a neutron is 0. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count... Then play a game to test your ideas!

Atom, smallest unit into which matter can be divided without the release of electrically charged particles. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz …. The tiny particles called atoms are the basic building blocks of all matter.

Charge and mass of neutrons.. This subatomic particle is located in the nuclei of atoms. The mass of a neutron is roughly equal to 1.008 atomic mass units. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The particle's mass is equal to approximately one atomic mass and, along with the atom's protons, accounts for the majority of the atom's overall mass. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Therefore, neutrons are neutrally charged subatomic particles. Charge and mass of neutrons.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Then play a game to test your ideas! Therefore, neutrons are neutrally charged subatomic particles. This subatomic particle is located in the nuclei of atoms. The tiny particles called atoms are the basic building blocks of all matter.. The tiny particles called atoms are the basic building blocks of all matter.

Electrons are extremely lightweight and exist in a cloud orbiting the nucleus... Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Then play a game to test your ideas!

As such, the atom is the basic building block of chemistry.. Identify the neutral atom described by name and mass number (i.e. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Charge and mass of neutrons... More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz …

The total number of these particles (called nucleons) in a given atom is called the mass number... Therefore, neutrons are neutrally charged subatomic particles. The electric charge that is associated with a neutron is 0. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. Identify the neutral atom described by name and mass number (i.e. Then play a game to test your ideas!. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons.

Atom, smallest unit into which matter can be divided without the release of electrically charged particles. Identify the neutral atom described by name and mass number (i.e. Answer each of the following using your knowledge of chemistry and the periodic table. As such, the atom is the basic building block of chemistry. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The symbol for proton number is the capital letter z. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.. It also is the smallest unit of matter that has the characteristic properties of a chemical element.

Then play a game to test your ideas! This subatomic particle is located in the nuclei of atoms. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. The total number of these particles (called nucleons) in a given atom is called the mass number. The symbol for proton number is the capital letter z. Answer each of the following using your knowledge of chemistry and the periodic table. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. The symbol for proton number is the capital letter z.

It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count... Therefore, neutrons are neutrally charged subatomic particles. It also is the smallest unit of matter that has the characteristic properties of a chemical element. As such, the atom is the basic building block of chemistry. The total number of these particles (called nucleons) in a given atom is called the mass number. The atomic mass of an atom is the sum of its protons and neutrons or z + n. Atom, smallest unit into which matter can be divided without the release of electrically charged particles.. Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change... Atoms can be combined with other atoms to form molecules , but they cannot be divided into smaller parts by ordinary means. The symbol for proton number is the capital letter z. The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. The total number of these particles (called nucleons) in a given atom is called the mass number. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Sep 11, 2019 · atomic particles. Identify the neutral atom described by name and mass number (i.e. The number of neutrons in an atom is indicated by the letter n. As such, the atom is the basic building block of chemistry... The atomic mass of an atom is the sum of its protons and neutrons or z + n.

Therefore, neutrons are neutrally charged subatomic particles. The atomic mass of an atom is the sum of its protons and neutrons or z + n.

Then play a game to test your ideas!. Then play a game to test your ideas!. Sep 11, 2019 · atomic particles.

Identify the neutral atom described by name and mass number (i.e... The electric charge that is associated with a neutron is 0. May 06, 2019 · while an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons.

The total number of these particles (called nucleons) in a given atom is called the mass number... Apr 25, 2017 · the neutron was discovered by james chadwick in 1932. Answer each of the following using your knowledge of chemistry and the periodic table. The large majority of an atom's mass comes from the protons and neutrons that make it up. More precisely, it is a characteristic of the system's total energy and momentum that is the same in all frames of reference related by lorentz … The number of neutrons in an atom is indicated by the letter n... It also is the smallest unit of matter that has the characteristic properties of a chemical element.