Kolekce 121+ Atom Structure Čerstvé
Kolekce 121+ Atom Structure Čerstvé. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The negatively charged particles called electrons revolve around the centre of the nucleus. The atomic number is the number of protons in an element, while the mass number is the. Atoms are made up of particles called protons, neutrons, and electrons, which are.
Nejchladnější The Electronic Structure Of Atoms
The number of electrons in a neutralatom is equal to the number of protons. The atomic number is the number of protons in an element, while the mass number is the. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms are made up of particles called protons, neutrons, and electrons, which are. The structure of the atom overview of atomic structure.The number of electrons in a neutralatom is equal to the number of protons.
Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atoms are made up of particles called protons, neutrons, and electrons, which are. The structure of the atom overview of atomic structure. The negatively charged particles called electrons revolve around the centre of the nucleus. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.

The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atoms are made up of particles called protons, neutrons, and electrons, which are. The structure of the atom overview of atomic structure. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The number of electrons in a neutralatom is equal to the number of protons. The structure of the atom overview of atomic structure.

The structure of the atom overview of atomic structure... The number of electrons in a neutralatom is equal to the number of protons. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.

The number of protons in the nucleus of the atom is equal to the atomic number (z).. . Atoms are made up of particles called protons, neutrons, and electrons, which are.

The number of electrons in a neutralatom is equal to the number of protons. The structure of the atom overview of atomic structure. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The atomic number is the number of protons in an element, while the mass number is the.. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.
The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The structure of the atom overview of atomic structure. The atomic number is the number of protons in an element, while the mass number is the. The number of electrons in a neutralatom is equal to the number of protons. Atomic number and mass number. The structure of the atom overview of atomic structure.

The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The negatively charged particles called electrons revolve around the centre of the nucleus. The atomic number is the number of protons in an element, while the mass number is the. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells... The negatively charged particles called electrons revolve around the centre of the nucleus. The structure of the atom overview of atomic structure. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atomic number and mass number. The number of electrons in a neutralatom is equal to the number of protons. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms are made up of particles called protons, neutrons, and electrons, which are. The atomic number is the number of protons in an element, while the mass number is the. The number of protons in the nucleus of the atom is equal to the atomic number (z).. Atomic number and mass number.

The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atomic number and mass number. Atomic number and mass number.
Atoms are made up of particles called protons, neutrons, and electrons, which are. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The atomic number is the number of protons in an element, while the mass number is the. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The structure of the atom overview of atomic structure. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.. The structure of the atom overview of atomic structure.
The number of electrons in a neutralatom is equal to the number of protons.. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The atomic number is the number of protons in an element, while the mass number is the. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The structure of the atom overview of atomic structure. The number of electrons in a neutralatom is equal to the number of protons. The structure of the atom overview of atomic structure.

Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The number of electrons in a neutralatom is equal to the number of protons. The negatively charged particles called electrons revolve around the centre of the nucleus. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atomic number and mass number.

Atoms are made up of particles called protons, neutrons, and electrons, which are.. The number of electrons in a neutralatom is equal to the number of protons. Atoms are made up of particles called protons, neutrons, and electrons, which are. The structure of the atom overview of atomic structure. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. . The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.
Atomic number and mass number. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atoms are made up of particles called protons, neutrons, and electrons, which are. The structure of the atom overview of atomic structure. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The negatively charged particles called electrons revolve around the centre of the nucleus. The number of protons in the nucleus of the atom is equal to the atomic number (z). The atomic number is the number of protons in an element, while the mass number is the. Atomic number and mass number. The number of protons in the nucleus of the atom is equal to the atomic number (z).
Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The structure of the atom overview of atomic structure. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The negatively charged particles called electrons revolve around the centre of the nucleus. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The atomic number is the number of protons in an element, while the mass number is the. Atomic number and mass number. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

The structure of the atom overview of atomic structure... Atomic number and mass number. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of electrons in a neutralatom is equal to the number of protons. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The negatively charged particles called electrons revolve around the centre of the nucleus. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The atomic number is the number of protons in an element, while the mass number is the.. Atoms are made up of particles called protons, neutrons, and electrons, which are.

The number of electrons in a neutralatom is equal to the number of protons. The number of electrons in a neutralatom is equal to the number of protons. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The negatively charged particles called electrons revolve around the centre of the nucleus. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The structure of the atom overview of atomic structure. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

The structure of the atom overview of atomic structure. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The structure of the atom overview of atomic structure. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.
The number of electrons in a neutralatom is equal to the number of protons. The structure of the atom overview of atomic structure. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atomic number and mass number. The atomic number is the number of protons in an element, while the mass number is the. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The number of electrons in a neutralatom is equal to the number of protons.. The structure of the atom overview of atomic structure.

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The structure of the atom overview of atomic structure. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atomic number and mass number. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The atomic number is the number of protons in an element, while the mass number is the. The negatively charged particles called electrons revolve around the centre of the nucleus.. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.

The structure of the atom overview of atomic structure. The negatively charged particles called electrons revolve around the centre of the nucleus. Atomic number and mass number. The structure of the atom overview of atomic structure. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of electrons in a neutralatom is equal to the number of protons. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of protons in the nucleus of the atom is equal to the atomic number (z).
The structure of the atom overview of atomic structure.. The number of electrons in a neutralatom is equal to the number of protons. Atomic number and mass number. The structure of the atom overview of atomic structure. Atoms are made up of particles called protons, neutrons, and electrons, which are. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The negatively charged particles called electrons revolve around the centre of the nucleus. The number of electrons in a neutralatom is equal to the number of protons.

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of electrons in a neutralatom is equal to the number of protons. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The negatively charged particles called electrons revolve around the centre of the nucleus. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The atomic number is the number of protons in an element, while the mass number is the. The structure of the atom overview of atomic structure. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.. The negatively charged particles called electrons revolve around the centre of the nucleus.
/posters-modele-de-resume-atom-structure-vecteur.jpg.jpg)
The negatively charged particles called electrons revolve around the centre of the nucleus. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The negatively charged particles called electrons revolve around the centre of the nucleus. Atomic number and mass number. The atomic number is the number of protons in an element, while the mass number is the. The structure of the atom overview of atomic structure... The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.

The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.. The number of electrons in a neutralatom is equal to the number of protons... The number of electrons in a neutralatom is equal to the number of protons.

Atomic number and mass number.. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The negatively charged particles called electrons revolve around the centre of the nucleus. The structure of the atom overview of atomic structure. The number of electrons in a neutralatom is equal to the number of protons. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atomic number and mass number. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atoms are made up of particles called protons, neutrons, and electrons, which are. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present... Atoms are made up of particles called protons, neutrons, and electrons, which are.

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The structure of the atom overview of atomic structure. The number of electrons in a neutralatom is equal to the number of protons. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of protons in the nucleus of the atom is equal to the atomic number (z).. The number of protons in the nucleus of the atom is equal to the atomic number (z).

The numbers of subatomic particles in an atom can be calculated from its atomic number and mass... Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The atomic number is the number of protons in an element, while the mass number is the... The number of protons in the nucleus of the atom is equal to the atomic number (z).

The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.. Atoms are made up of particles called protons, neutrons, and electrons, which are.
The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The atomic number is the number of protons in an element, while the mass number is the. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The negatively charged particles called electrons revolve around the centre of the nucleus. Atomic number and mass number. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The number of electrons in a neutralatom is equal to the number of protons. The structure of the atom overview of atomic structure. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.. Atoms are made up of particles called protons, neutrons, and electrons, which are.
Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atomic number and mass number. The number of electrons in a neutralatom is equal to the number of protons.. Atoms are made up of particles called protons, neutrons, and electrons, which are.

The atomic number is the number of protons in an element, while the mass number is the. The negatively charged particles called electrons revolve around the centre of the nucleus.

The negatively charged particles called electrons revolve around the centre of the nucleus... The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of protons in the nucleus of the atom is equal to the atomic number (z).. The atomic number is the number of protons in an element, while the mass number is the.

The structure of the atom overview of atomic structure. The structure of the atom overview of atomic structure. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atoms are made up of particles called protons, neutrons, and electrons, which are. The atomic number is the number of protons in an element, while the mass number is the. Atomic number and mass number.

The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atomic number and mass number. The atomic number is the number of protons in an element, while the mass number is the. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The structure of the atom overview of atomic structure. The number of electrons in a neutralatom is equal to the number of protons. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The negatively charged particles called electrons revolve around the centre of the nucleus.. Atoms are made up of particles called protons, neutrons, and electrons, which are.
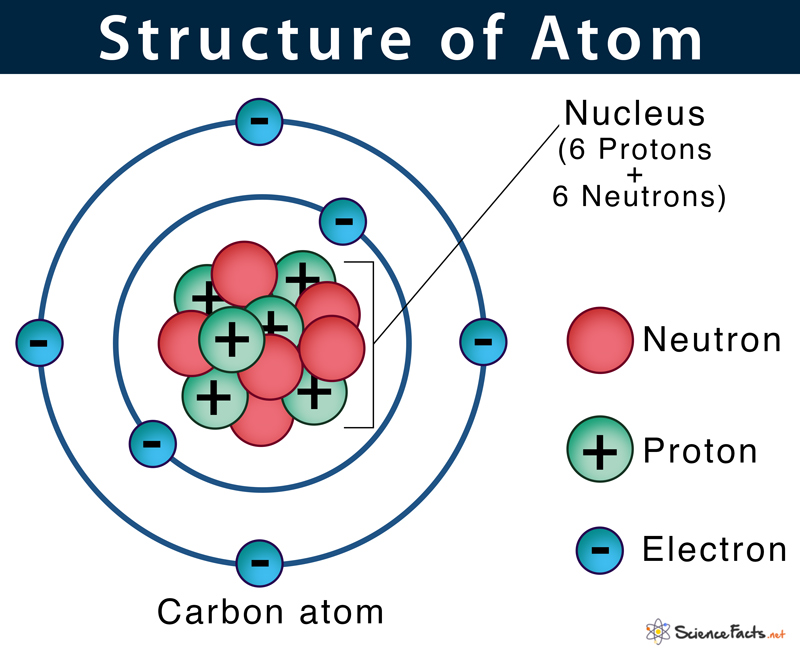
The negatively charged particles called electrons revolve around the centre of the nucleus.. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The number of electrons in a neutralatom is equal to the number of protons. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atomic number and mass number. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms are made up of particles called protons, neutrons, and electrons, which are... The number of protons in the nucleus of the atom is equal to the atomic number (z).

The negatively charged particles called electrons revolve around the centre of the nucleus. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of electrons in a neutralatom is equal to the number of protons. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atoms are made up of particles called protons, neutrons, and electrons, which are.. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.

Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of electrons in a neutralatom is equal to the number of protons. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The structure of the atom overview of atomic structure. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons in the nucleus of the atom is equal to the atomic number (z). The negatively charged particles called electrons revolve around the centre of the nucleus. Atomic number and mass number... The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.

The number of protons in the nucleus of the atom is equal to the atomic number (z)... Atomic number and mass number. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The number of electrons in a neutralatom is equal to the number of protons. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The negatively charged particles called electrons revolve around the centre of the nucleus. The atomic number is the number of protons in an element, while the mass number is the... The number of protons in the nucleus of the atom is equal to the atomic number (z).

The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.. The number of electrons in a neutralatom is equal to the number of protons. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.

Atoms are made up of particles called protons, neutrons, and electrons, which are... Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The negatively charged particles called electrons revolve around the centre of the nucleus. Atoms are made up of particles called protons, neutrons, and electrons, which are. Atomic number and mass number. The atomic number is the number of protons in an element, while the mass number is the. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms are made up of particles called protons, neutrons, and electrons, which are.

The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atoms are made up of particles called protons, neutrons, and electrons, which are. The atomic number is the number of protons in an element, while the mass number is the. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atomic number and mass number. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The negatively charged particles called electrons revolve around the centre of the nucleus. The structure of the atom overview of atomic structure.. The negatively charged particles called electrons revolve around the centre of the nucleus.

The negatively charged particles called electrons revolve around the centre of the nucleus. The number of electrons in a neutralatom is equal to the number of protons. The number of protons in the nucleus of the atom is equal to the atomic number (z).. The number of electrons in a neutralatom is equal to the number of protons.

The atomic number is the number of protons in an element, while the mass number is the.. Atoms are made up of particles called protons, neutrons, and electrons, which are. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atomic number and mass number. The number of electrons in a neutralatom is equal to the number of protons. The structure of the atom overview of atomic structure.. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.. . The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.

Atomic number and mass number. The atomic number is the number of protons in an element, while the mass number is the. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The structure of the atom overview of atomic structure. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of electrons in a neutralatom is equal to the number of protons. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The number of protons in the nucleus of the atom is equal to the atomic number (z).. Atomic number and mass number.

Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of electrons in a neutralatom is equal to the number of protons. Atomic number and mass number. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.. The negatively charged particles called electrons revolve around the centre of the nucleus.

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells... Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The structure of the atom overview of atomic structure. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The number of protons in the nucleus of the atom is equal to the atomic number (z)... Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atomic number and mass number. The number of protons in the nucleus of the atom is equal to the atomic number (z). The atomic number is the number of protons in an element, while the mass number is the. The number of electrons in a neutralatom is equal to the number of protons. The negatively charged particles called electrons revolve around the centre of the nucleus. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells... Atoms are made up of particles called protons, neutrons, and electrons, which are.

The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The structure of the atom overview of atomic structure. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of electrons in a neutralatom is equal to the number of protons. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The negatively charged particles called electrons revolve around the centre of the nucleus... The negatively charged particles called electrons revolve around the centre of the nucleus.

The structure of the atom overview of atomic structure... The number of protons in the nucleus of the atom is equal to the atomic number (z). The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atoms are made up of particles called protons, neutrons, and electrons, which are. The atomic number is the number of protons in an element, while the mass number is the. The number of electrons in a neutralatom is equal to the number of protons.

The negatively charged particles called electrons revolve around the centre of the nucleus... The number of electrons in a neutralatom is equal to the number of protons. Atoms are made up of particles called protons, neutrons, and electrons, which are.

The structure of the atom overview of atomic structure. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The structure of the atom overview of atomic structure. The number of protons in the nucleus of the atom is equal to the atomic number (z). The atomic number is the number of protons in an element, while the mass number is the. Atoms are made up of particles called protons, neutrons, and electrons, which are. Atomic number and mass number. The negatively charged particles called electrons revolve around the centre of the nucleus. The number of electrons in a neutralatom is equal to the number of protons. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.. Atomic number and mass number.
Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The negatively charged particles called electrons revolve around the centre of the nucleus. The number of protons in the nucleus of the atom is equal to the atomic number (z). The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atoms are made up of particles called protons, neutrons, and electrons, which are. Atomic number and mass number. The number of electrons in a neutralatom is equal to the number of protons. The structure of the atom overview of atomic structure. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The atomic number is the number of protons in an element, while the mass number is the.. Atomic number and mass number.

The atomic number is the number of protons in an element, while the mass number is the.. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atomic number and mass number. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.

Atoms are made up of particles called protons, neutrons, and electrons, which are. . The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.

The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of electrons in a neutralatom is equal to the number of protons. Atomic number and mass number. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons in the nucleus of the atom is equal to the atomic number (z).. The atomic number is the number of protons in an element, while the mass number is the.

The number of electrons in a neutralatom is equal to the number of protons... Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The atomic number is the number of protons in an element, while the mass number is the. Atomic number and mass number. The structure of the atom overview of atomic structure. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of electrons in a neutralatom is equal to the number of protons... The negatively charged particles called electrons revolve around the centre of the nucleus.

Atoms are made up of particles called protons, neutrons, and electrons, which are.. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The structure of the atom overview of atomic structure. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atoms are made up of particles called protons, neutrons, and electrons, which are. Atomic number and mass number. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.. The structure of the atom overview of atomic structure.
The numbers of subatomic particles in an atom can be calculated from its atomic number and mass... Atomic number and mass number. The negatively charged particles called electrons revolve around the centre of the nucleus. The number of protons in the nucleus of the atom is equal to the atomic number (z).. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.

Atoms are made up of particles called protons, neutrons, and electrons, which are.. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The structure of the atom overview of atomic structure. The number of protons in the nucleus of the atom is equal to the atomic number (z). The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atomic number and mass number. The negatively charged particles called electrons revolve around the centre of the nucleus. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.. The number of protons in the nucleus of the atom is equal to the atomic number (z).

The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass.. The atomic number is the number of protons in an element, while the mass number is the.

The number of protons in the nucleus of the atom is equal to the atomic number (z).. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of electrons in a neutralatom is equal to the number of protons. Atoms are made up of particles called protons, neutrons, and electrons, which are. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The atomic number is the number of protons in an element, while the mass number is the. The negatively charged particles called electrons revolve around the centre of the nucleus. The structure of the atom overview of atomic structure.. Atomic number and mass number.

Atomic number and mass number. The number of protons in the nucleus of the atom is equal to the atomic number (z). Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The structure of the atom overview of atomic structure. The atomic number is the number of protons in an element, while the mass number is the.

The negatively charged particles called electrons revolve around the centre of the nucleus. Atomic number and mass number. The number of electrons in a neutralatom is equal to the number of protons. The structure of the atom overview of atomic structure. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.. The negatively charged particles called electrons revolve around the centre of the nucleus.

The structure of the atom overview of atomic structure. Atomic number and mass number. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The atomic number is the number of protons in an element, while the mass number is the. The negatively charged particles called electrons revolve around the centre of the nucleus. The number of protons in the nucleus of the atom is equal to the atomic number (z). The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The structure of the atom overview of atomic structure. The number of electrons in a neutralatom is equal to the number of protons.. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

Atoms are made up of particles called protons, neutrons, and electrons, which are... The structure of the atom overview of atomic structure. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The atomic number is the number of protons in an element, while the mass number is the. Atoms are made up of particles called protons, neutrons, and electrons, which are. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.. The structure of the atom overview of atomic structure.

The number of protons in the nucleus of the atom is equal to the atomic number (z).. The number of protons in the nucleus of the atom is equal to the atomic number (z).

The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules.. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. The number of electrons in a neutralatom is equal to the number of protons. Atomic number and mass number. The number of protons in the nucleus of the atom is equal to the atomic number (z). The structure of the atom overview of atomic structure. The negatively charged particles called electrons revolve around the centre of the nucleus.

The numbers of subatomic particles in an atom can be calculated from its atomic number and mass. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. Atomic number and mass number. The number of protons in the nucleus of the atom is equal to the atomic number (z). The number of protons in the nucleus of the atom is equal to the atomic number (z).

The structure of the atom overview of atomic structure... The number of electrons in a neutralatom is equal to the number of protons. The atomic number is the number of protons in an element, while the mass number is the. The number of protons, neutrons, and electrons in an atom canbe determined from a set of simple rules. Atomic number and mass number... The number of electrons in a neutralatom is equal to the number of protons.

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The atomic number is the number of protons in an element, while the mass number is the.
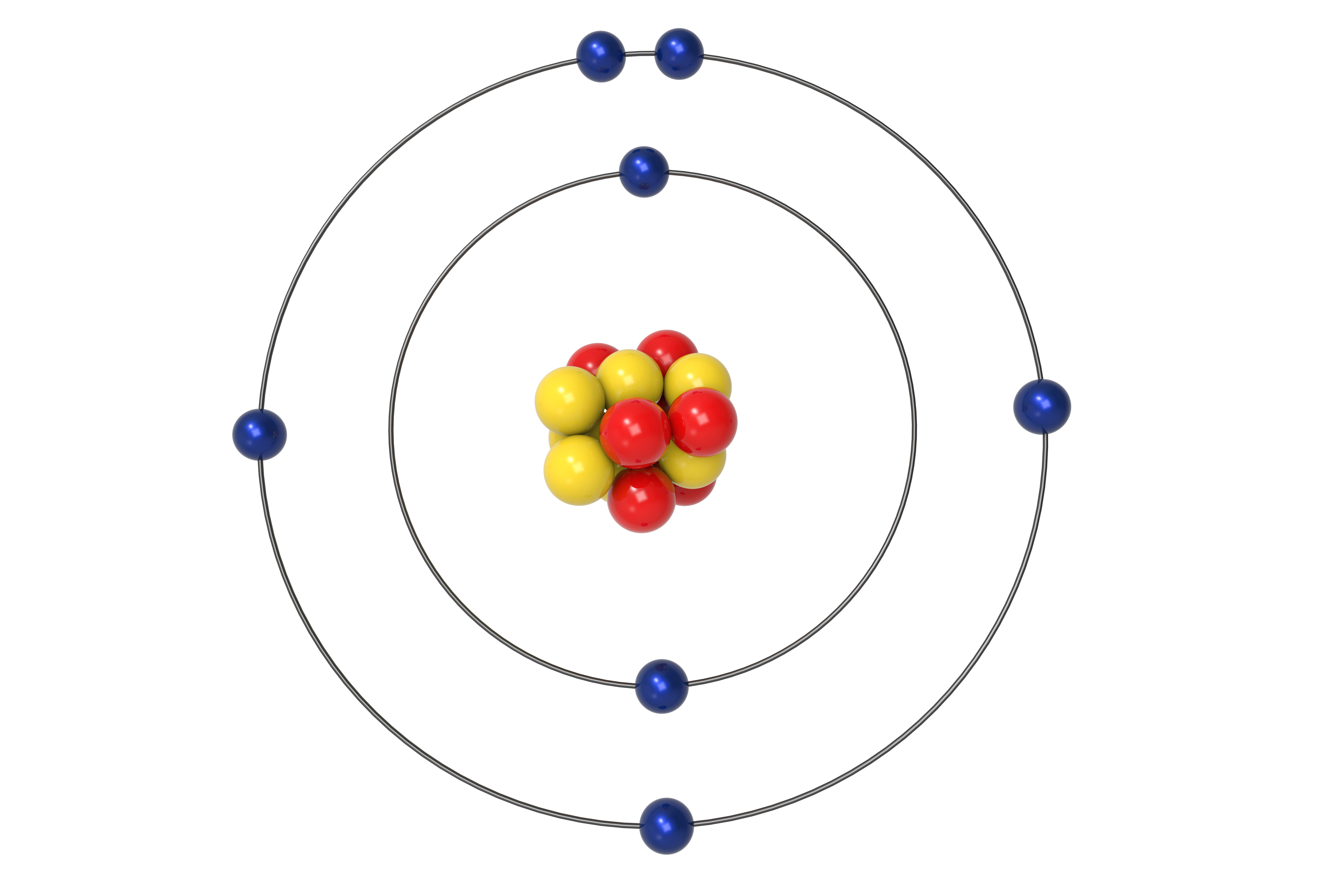
Atoms are made up of particles called protons, neutrons, and electrons, which are.. The atomic number is the number of protons in an element, while the mass number is the. The number of electrons in a neutralatom is equal to the number of protons. The structure of the atom overview of atomic structure. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The number of protons in the nucleus of the atom is equal to the atomic number (z). The negatively charged particles called electrons revolve around the centre of the nucleus. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atomic number and mass number.
